 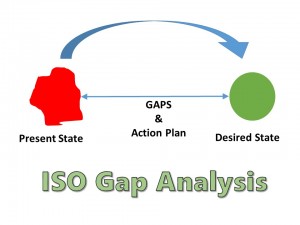
Pre- and post- accreditation audits of nonconformities for management and technical requirements of the ISO/IEC17025:2005 standards were evaluated from a Quality Control laboratory in the National Agency for Food and Drug Administration and Control (NAFDAC), located in Nigeria, West Africa. This study investigated the impact of ISO/IEC 17025 accreditation on the performance of an NMRA Quality Control laboratory to provide evidence of improved quality compliance within a low-resource setting. Sharing the benefits of laboratory accreditation could help more countries within sub-Saharan Africa overcome existing challenges to achieve accreditation and robust quality systems. Although countries desire to have reliable medicine testing facilities, accrediting a national laboratory to international standards poses financial and technical challenges for many low-income countries. The National Medicines Regulatory Authority (NMRA) Quality Control laboratories assess the quality of medicines used by the populace as part of their regulatory function. Quality standards are established and maintained to ensure the reliability of laboratory test reports. The number and severity of nonconformities generated during an audit of a medicine testing laboratory indicates its level of quality compliance. According to the outcomes, the company was mostly compliant with the standard, and there was a need for slight modifications or updates in the system to be fully conformed. It gives a quantitative approach to the research. As well as this study use gap analysis techniques compare to the existing situations with the expected conditions. 
The secondary data was collected from reliable sources of information, including guidebooks and standards linked to the study. The key data was collected through questionnaires, interviews, observations and study of internal documents. The main objectives of this study are to identify and analyze the gaps and also implement the ISO/IEC 17025:2017 quality management system in conformance with the standard. Those are the requirements of ISO/IEC 17025:2017 standard, the construction of a quality manual, and also the company's readiness for accreditation. Quality is a comprehensive topic, and it indeed cannot be covered thoroughly within this project, therefore focused on three significant angles that could help the process of implementing quality systems become easier. A company with good quality practices simply increases faith and interest from existing customers and also from potential customers. Having a quality management system is part of strategic business development. Implementation and improvement of the quality management system is an objective of many companies. This instruction will assure the testing laboratory to carry out testing services consistently and reliably. 
ISO/IEC Guide 17025 is a universal guideline that comprises benchmarks for product testing service laboratories. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
